|
|
BMe Research Grant |
|
Doctoral School of Psychology (Cognitive Science)
Department of Cognitive Sciences
Supervisor: Dr. Winkler István
Individual Differences in Perceiving Multistable Sound Sequences
Introduction of the research area
Perceptual bi- or multistabilities or perceptual illusions are used to study perception from the beginning of perception science. These perceptual configurations do not have one good solution which can be accepted by the perceptual system, thus it is constantly switching between the possible interpretations of the stimuli. Knowledge on normal perception can also be acquired through mapping these weaknesses of the perceptual system. Conscious perceptual experience in multistabilities depends on the peculiarity of the perceiver’s own nervous system. Thus, multistable perception can be used to gain more knowledge on differences between individuals. Focusing on this second possibility, in my research I would like to gain more insight into the dimensions of individual differences in perception using an auditory multistability paradigm, psychological tests, and neuroscientific techniques.
Brief introduction of the research place
My research is conducted under the supervision of Prof. Dr. István Winkler at the Institute of Cognitive Neuroscience and Psychology, Research Centre for Natural Sciences, Hungarian Academy of Sciences. Our research group’s main profile is auditory research with cognitive and neuroscientific methods. My research is done under a Momentum grant aimed to understand the psychology of auditory scene analysis more deeply.
History and context of the research
In the sensory phenomenon of bi/multistable perception the conscious perceptual awareness switches stochastically between possible interpretations of an unchanging stimulus (see Schwartz et al., 2012). One of the most well-known perceptual bistability is the duck-rabbit illusion (Figure 1), where the perceiver can view the image as a duck or a rabbit and perception switches between the two possible interpretations. In the study of perceptual bi- and multistabilities, it is known for quite a long time (Aafjes et al., 1966) that there is high individual variability in the switches between the alternative interpretations. In other words, some individuals switch more, whereas others switch less.

Figure 1. The duck-rabbit illusion (1892): an example of a visual bistability.
Denham and her colleagues (2014) described perceivers’ overall switching pattern beyond the number of switches using an auditory multistability paradigm called auditory streaming. Participants listened to a sound sequence of alternating low-high-low-break sounds and they were constantly indicating their current percept from the available alternatives (Figure 2). Based on this data, the switching pattern contains the information on the probability of holding on to a perceptual pattern or switching to another one. Using this richer characterization of the switching pattern compared to the number of switches, the authors found that listeners have an idiosyncratic switching pattern compared to the other participants. A year later after the first experiment, the same listeners retained their switching pattern. Thus, it seems that these individual differences have high temporal stability.
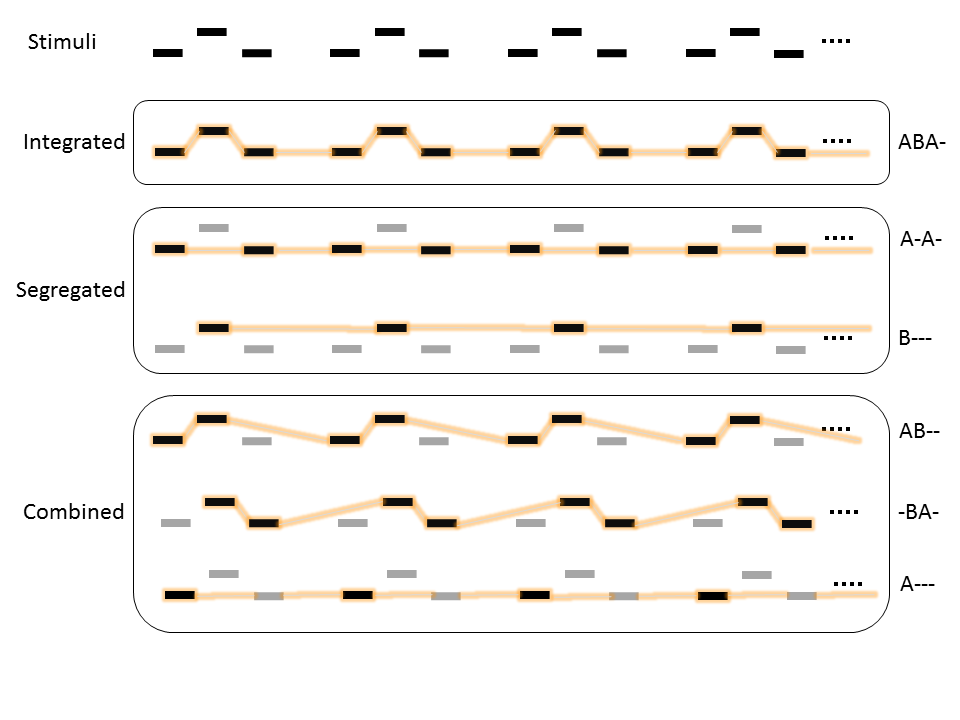
Figure 2. The possible perceptual patterns and their categories in the auditory streaming paradigm during a continuous presentation of low-high-low-break (ABA-) sound sequence. Dominant streams are emphasized with orange.
Research goal, open questions
The ultimate aim of my research is to explore the background of the individual differences in the switching patterns to gain more insight into perception and personality. A first set of questions concerns the possible correlates from various psychological constructs with the individual differences in the auditory streaming paradigm. Such constructs are executive functions responsible for the management of cognitive processes like inhibition, shifting and working memory. Furthermore, personality dimensions linked with these individual differences are aimed to be explored. There is no available preliminary information on these topics, thus it is possible that these differences are affected by openness, agreeableness, impulsivity or flexibility. The actual behavior stems from the nervous activation, thus the second set of questions is centered on indicators on the level of the brain, which can predict individual characteristics of perceptual multistabilities.
Methods
All of my studies presented here used the auditory streaming paradigm. First, participants are trained to match their perceptual experiences to the appropriate buttons, and later during the experiment, they are continuously indicating the perceptual pattern they hear at the moment. Individual differences are defined as switching patterns constructed from four four-minute-long blocks (Farkas et al., under review). Every individual can be described with a switching pattern. Distances between participants’ switching patterns are calculated and visualized in a two dimensional space. These dimensions are interpreted with so called directly observable perceptual variables, namely the average durations and proportions of each perceptual pattern, the average number of switches and the time required to discover all possible patterns.
Components of the executive functions are measured with separate tasks, whereas personality is measured with scales. From these scales the ego-resiliency scale (Block, 2002, Farkas & Orosz, 2015) should be highlighted, which measures the flexibility of personality. Individuals with high levels of ego-resiliency possess the skills that make them solve everyday issues and situations adaptively. They are also characterized by an active openness to the world.
The following two methods are used to measure activation in the brain. The first is the electroencephalogram (EEG), where the electric activity of the brain is registered with electrodes attached to the scalp. The other one is the possibly less known and used Near-Infrared Spectroscopy (NIRS). The basic principle of this method is that near-infrared light goes through biological tissues. For example, this is the reason why our hand becomes red when we put a flashlight under it. In the near-infrared region, the oxygenated and deoxygenated hemoglobins reflect light differently (Figure 3). Thus, with light emitted from sources placed on the head are reflected from the hemoglobins, which can be registered with detectors also placed in the scalp (Figure 4). This method allows us to calculate the concentration changes of oxygenated and deoxygenated hemoglobins in the upper layers of the cortex. Activation in a brain region is expected to elicit an increased concentration of oxygenated and a decreased concentration of deoxygenated hemoglobins.
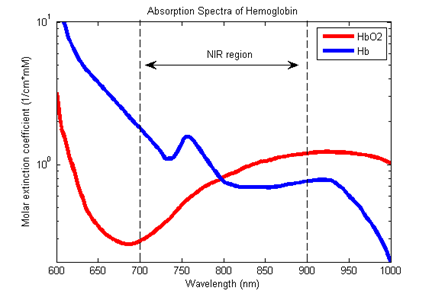
Figure 3. In the near-infrared region (wavelengths between 700–900 nanometers), the oxygenated (HbO2, red) and deoxygenated (Hb, blue) hemoglobins reflect light differently.
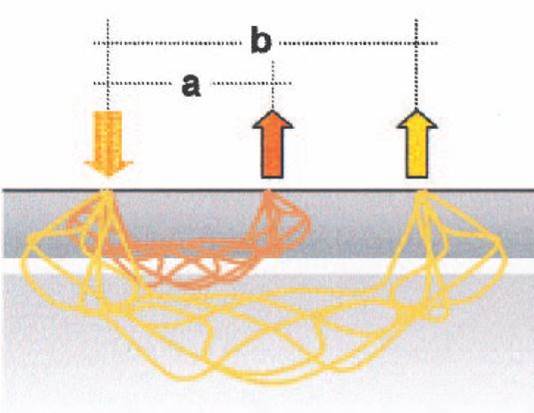
Figure 4. If source-detector pairs are too close to each other (distance ‘a’), only irrelevant information is registered from the skull. In case of an appropriate distance (2–3 cm, ‘b’), the extent of the scattering of light is acceptable and the blood-related concentration changes can be measured (Obrig & Villringer, 2003).
NIRS data are mainly analyzed in the framework of functional connectivity. With functional connectivity it is possible to map relevant hubs and their network through the temporal correlation of activation in certain brain regions or measurement channels in case of the NIRS (Bullmore & Sporns, 2009). Graph theory is used to create an abstract representation of these networks. Graphs are calculated for every participant with the Minimum Spanning Tree algorithm, which does not allow closed structures within a graph (Figure 5). Then graphs are described with various indices, such as hierarchy or degree (Stam & van Straaten, 2012). These indices can be used in group-level analysis to identify individual characteristics of the brain activation in the auditory streaming paradigm and study the possible causes of individual differences.
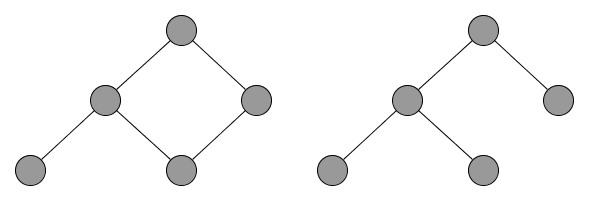
Figure 5. Example of simple graphs: the Minimum Spanning Tree algorithm produces graphs without closed structures as can be seen on the right side of the figure.
Results
Two experiments were conducted in this topic. The first one focused on psychological constructs that can be possibly linked to individual differences in the switching pattern. Beyond the executive functions of inhibition, shifting, and working memory, multiple personality inventories were administered including the ego-resiliency scale. Creativity was also assessed. 87.5% of the participants had an idiosyncratic switching pattern in this experiment. The main dimension which individuals differed from each other was named Exploration, because on one end of this dimension there were participants who experienced the least frequent perceptual pattern more, the most frequent perceptual patterns less, switched more between patterns, and required less time to discover all possible interpretations of the stimuli than participants at the other end of the dimension. From the psychological constructs listed above, only ego-resiliency showed a reliable correlation with the Exploration dimension in a way that high ego-resiliency was associated with more Exploration.
In the second experiment the emphasis was placed on neuroscientific methods: both EEG and NIRS were registered during the experiment, and ego-resiliency was registered again. In this experiment, 85.29% of the participants had an idiosyncratic switching pattern and the distances between them were described by the same dimensions. Moreover, the result of the correlation between ego-resiliency and Exploration was also replicated. After preprocessing the NIRS data of the changes in the deoxygenated hemoglobin concentration during the multistable auditory task, every participant's graph was constructed and described with various indicators. It seems that the hierarchy of the networks is related to the dimension of Exploration. For the sake of a clearer understanding, the graphs of the most and least explorative 25% of the participants were constructed separately (Figure 6), and different brain regions were depicted with different colors. The network of the less explorative individuals is hierarchic: the connections are grouped to three main hubs. The first one is a frontal (green), which is responsible for the management of the information, the second one is a parietal (yellow), which is responsible for the integration of information, and a right temporal one (blue), which is responsible for the processing of auditory stimuli. The three hubs communicate with each other through a fronto-parietal pathway. The network of the more explorative individuals has more hubs with fewer connections. It is important to note that the direction of how the information travels between the hubs cannot be explored from these figures.
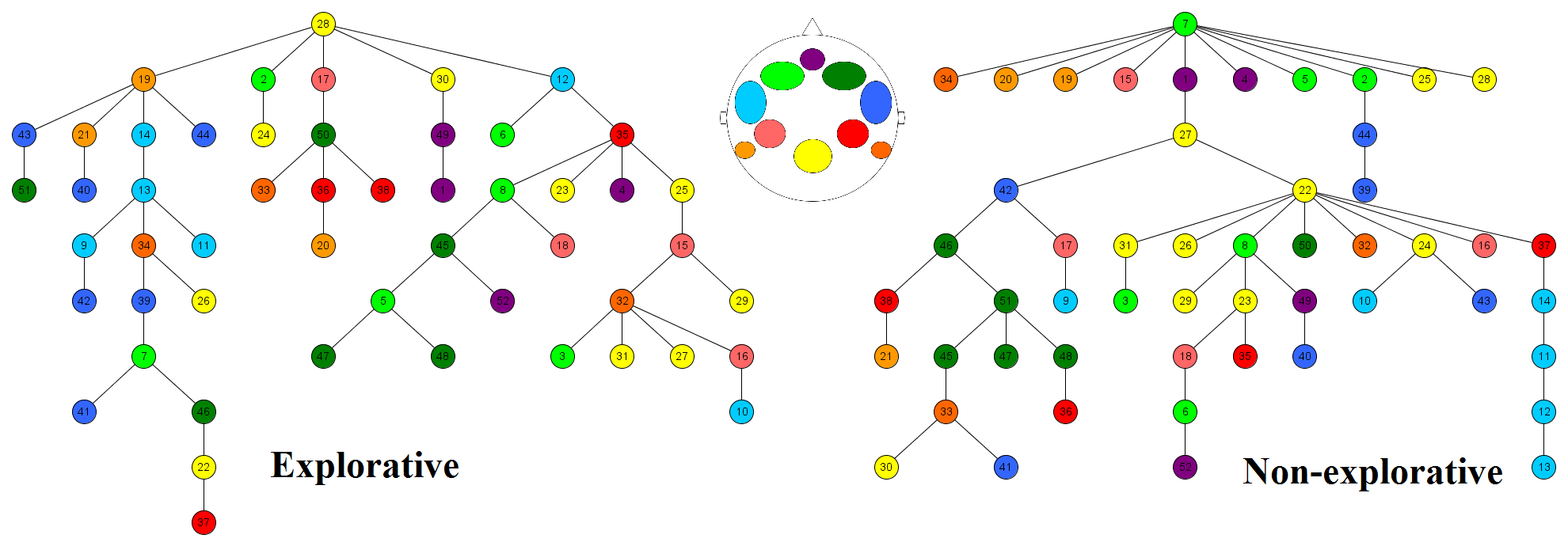
Figure 6. Networks of the explorative and not explorative individuals. Color codes of the regions: purple – prefrontal, green – frontal, blue – anterior auditory cortex, red – anterior parietal, red – anterior parietal, orange – posterior auditory cortex, yellow – posterior parietal. Hemispheric differences are marked with different shades within a color.
Expected impact and future research
One of my short term aims is to map the networks activated during the perception of each perceptual patterns with graphs. These will serve as an important tool to interpret individual networks. After that I plan to analyze EEG data similarly. EEG and NIRS measure qualitatively different information, thus results supported by both methods and results specific to method will provide valuable information.
My long term goal is to compare auditory multistabilities with visual ones to investigate the specificity of brain networks and individual differences to a certain modality. The focus of most of the research on multistabilities or perception in general is not on individual differences. Thus, every study concerning them is important in the field, especially if they provide information on the neural background.
Publications, references, links
Related publications
Farkas, D., Denham, S. L., Bendixen, A., & Winkler, I. (under review). Assessing the validity of subjective reports in the auditory streaming paradigm. The Journal of the Acoustical Society of America.
Farkas, D., & Orosz, G. (2015). Ego-Resiliency Reloaded: A Three-Component Model of General Resiliency. PloS one, 10(3), e0120883.
References
Aafjes, M., Hueting, J. E., & Visser, P. (1966). Individual and interindividual differences in binocular rivalry in man. Psychophysiology, 3, 18–22.
Block, J. (2002). Personality as an affect-processing system: Toward an integrative theory. Psychology Press.
Bullmore, E., & Sporns, O. (2009). Complex brain networks: graph theoretical analysis of structural and functional systems. Nature Reviews Neuroscience, 10(3), 186–198.
Denham, S. L., Bőhm, T. M., Bendixen, A., Szalárdy, O., Kocsis, Z., Mill R., and Winkler, I. (2014). „Stable individual characteristics in the perception of multiple embedded patterns in multistable auditory stimuli”, Front. Neurosci. 8(25). doi:10.3389/fnins.2014.00025.
Obrig, H., & Villringer, A. (2003). Beyond the visible—imaging the human brain with light. Journal of Cerebral Blood Flow & Metabolism, 23(1), 1–18.
Schwartz, J.-L., Grimault, N., Hupé, J.-M., Moore, B. C. J., & Pressnitzer, D. (2012) Multistability in perception: Binding sensory modalities, an overview. Philosophical Transaction Royal Society of London B, 367, 896–905.
Stam, C.J., & van Straaten, E.C. (2012). The organization of physiological brain networks. Clinical Neurophysiology, 123, 1067–108 7.
